In particular, reported N1s XPS peaks at 400–402 eV, assigned as quaternary N (Q-N), tertiary amine (T–N), or graphitic N, have been still ambiguous until now because the meaning of graphitic N was uncertain whether graphitic N is Q–N or T–N, and the presence of Q–N was uncertain for most carbon materials with complexed structures. Although the types of N-containing functional groups have been generally revealed by XPS owing to the effort of researchers, misinterpretations of assignments of N1s XPS spectra have been pointed out including our previous works. X-ray photoelectron spectroscopy (XPS) is among the most powerful and essential techniques to analyse the structures of N-doped carbon materials qualitatively and quantitatively. Thus, clarification of the bonding state of graphitic N is essential for the research of N-doped carbon materials. Graphitic N or substitutional N in the basal plane of carbon materials is especially an important functional group because of the stability compared to pyridinic and pyrrolic N, which leads to the high cyclic performance as electrodes and stable active sites for catalytic reactions. Nitrogen (N)-doped carbon materials such as graphene, carbon nanotube, graphene nanoribbon, carbon fiber, and activated carbon have attracted attention because of the high performance for various applications such as fuel cell and electric double-layer capacitor.
Besides, it was revealed that the peak at 400.1 eV originated from T–N on 5-membered rings or 7- and 5-membered rings, but not pyrrolic N because graphite including no hydrogen was used for analysis. The peaks at 400.1–401.2 eV were determined to be T–N, but not Q–N by analyzing graphitized polyimide (with the oxygen content of 0.01 at% or lower and the hydrogen content of 0 at%) using Raman spectroscopy, XPS, X-ray diffraction, total neutron scattering, elemental analysis, and molecular dynamics simulation. In this work, it was clarified that one of the reasons for the prevailing ambiguous assignments is the presence of N in heptagonal and pentagonal rings. 400 eV has been assigned as pyrrolic N, but the presence of N–H is generally not confirmed. 401 eV as Q–N or graphitic N, whereas raw materials in most of those works contain neither counter anion nor halogen.
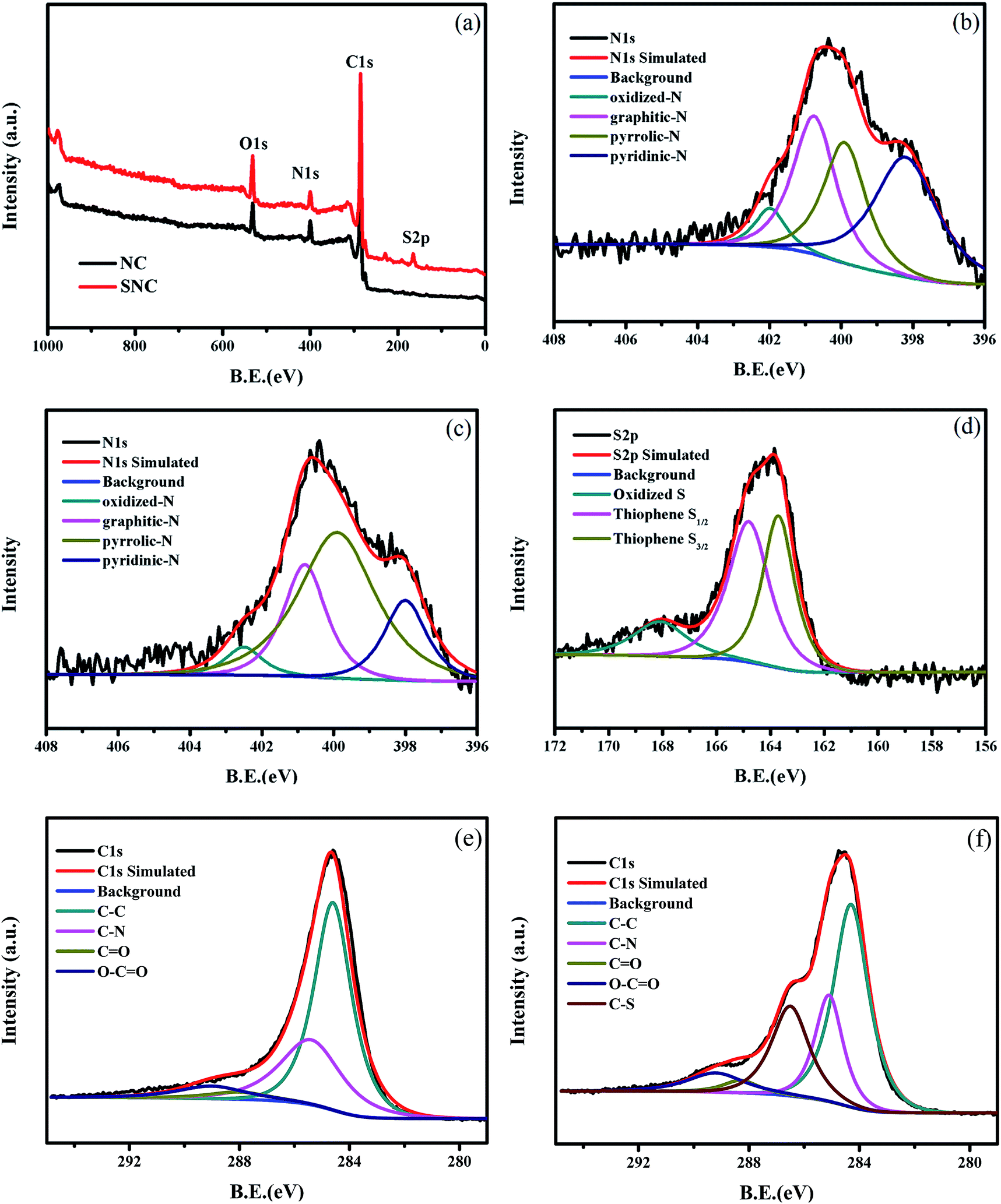
However, reported assignments of (1) graphitic nitrogen (N)/substitutional N, quaternary N (Q–N), or tertiary amine (T–N) and (2) pyrrolic N/secondary amine or T–N are questionable. X-ray photoelectron spectroscopy (XPS) is among the most powerful techniques to analyse structures of nitrogen-doped carbon materials.
